Hydrogen in a quantum hexagonal lattice may be a subatomic matrix for life and clay may be a next step up as a crystalline matrix for protein formation.
Long before life as we know it emerged, tiny clay crystals might have been the first spark of life on Earth, according to a bold scientific idea. Imagine these clay minerals, formed from the Earth’s rocky crust, acting like primitive living entities. Their intricate structures, with defects or random patterns in their crystal lattices, could have served as a kind of “genetic code,” much like the DNA in modern cells. These clays could “replicate” by breaking apart, passing on their unique patterns to new crystals. Over time, the most stable patterns thrived, while less stable ones dissolved away, in a process resembling natural selection. Eventually, these clay-based “life forms” may have provided a scaffold for organic molecules—the building blocks of life—to organize and evolve into the complex biochemistry we see today. Scientists like Cairns-Smith, who pioneered this idea, and others like Greenwell and Coveney, who suggested similar roles for related minerals called layered double hydroxides, propose that these minerals stored and transferred information in a way that kickstarted life.
To picture how clay minerals might have been the first “living” entities, imagine a mosaic of interlocking puzzle pieces or a tessellation artwork, like an intricate tile pattern. Each piece represents a tiny crystal of clay, with a unique arrangement of defects—think of these as slight imperfections or irregularities in the crystal’s structure, like a missing or misplaced tile in the mosaic. These defects form a distinct pattern, much like a primitive “genetic code,” similar to how DNA’s sequence of molecules carries information in modern cells.
When a clay crystal “replicates,” it’s as if the puzzle piece snaps apart, and each fragment serves as a template for new pieces to form, carrying forward the same pattern of defects. Just as tessellation art repeats a motif to create a larger design, these clay crystals could grow and split, passing their unique patterns to new crystals. This process, driven by the natural growth and dissolution of clay particles, might have allowed the most stable patterns to persist and evolve, laying the groundwork for life’s chemistry. Earth’s minerals might have played a starring role in the origin of life.
The clay template theory suggests that the intricate structures of clay minerals, particularly their crystal lattice defects, aperiodic cation distributions, or random layer stacking sequences, acted as a primitive form of genetic information. These patterns within the clay crystals could “replicate” when the crystals cleave or break apart during growth. The key mechanism is that each fragment retains a similar pattern to the original, allowing it to serve as a template for further crystal growth. This process favors the most stable or “fit” patterns, which persist and evolve through selective growth and dissolution, much like natural selection.

However, it’s not quite that each tiny bit of clay contains the “whole” in a holographic sense—where every piece encodes the entire structure, as in a hologram. Instead, the replication is more about the pattern itself being copied and propagated.
The theory by Hazen and Sverjensky, 2010, leans more toward a pattern-replication model, where the local arrangement of defects or layers in a clay fragment resembles the parent crystal’s pattern and can influence the growth of new crystals.
The unique texture of clay crystals, with their intricate defects and patterns, may have acted like a natural scaffold, holding amino acids in specific positions to encourage the formation of early protein-like chains. Think of it as a primitive version of a ribosome, which uses messenger RNA to guide protein assembly—except here, the clay’s structure served as a template, subtly directing the arrangement of life’s building blocks rather than encoding a precise sequence. This supportive role of mineral surfaces, including silicates like clay, has been observed in experiments where they concentrate and stabilize amino acids and other molecules, a key step toward life’s chemistry. It’s a bit like the grit you might find stuck in a kitchen floor crevice—over time, that stubborn residue can transform into a new, hard-to-remove substance, hinting at how Earth’s minerals might have catalyzed the stubborn chemistry of early life.
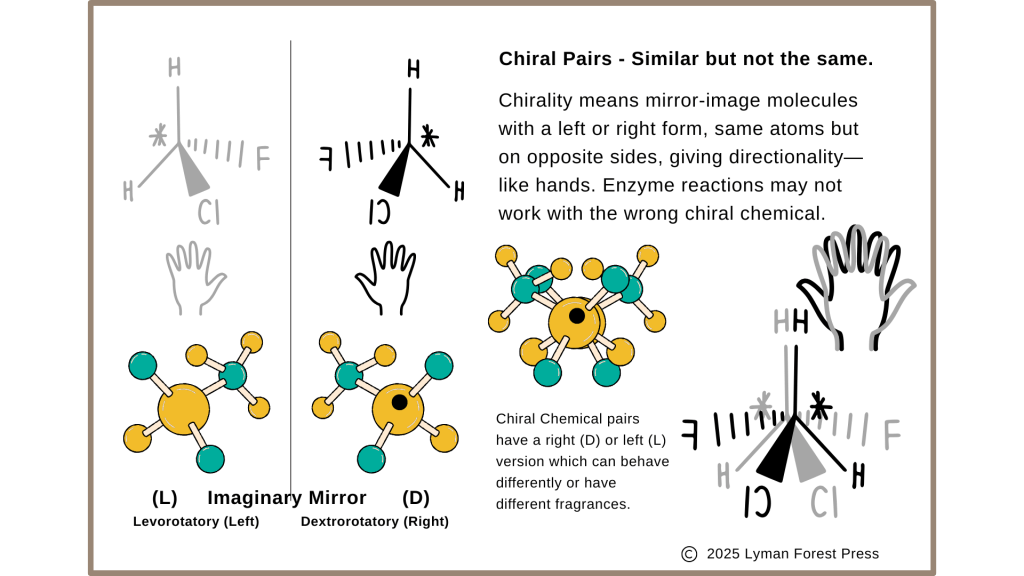
Silicon or clay can provide a template for assembly of molecules from smaller elements or molecules that collect on the surface. Some molecules have mirror images making them chemically similar but opposite in alignment. Our right and left hands are mirror images of each other. Chirality of molecules affects how likely or in what direction the alignment will occur on a crystalline surface like a drying clay mud puddle. The universe seems to prefer Left, L-amino acids, over Right, R-amino acids. Why is that? Possibly because the hydrogen quantum phase prefers Left chirality.

Chiral Pairs – Mirror Image Chemicals (L) and (D)
Beyond this, clay minerals and other common rock-forming minerals, like oxides, silicates, and carbonates, likely played a key role in setting the stage for life. Their surfaces acted like natural laboratories, gathering and concentrating essential molecules such as amino acids, sugars, and lipids—the raw materials of life. These surfaces could stabilize these molecules, protecting them from breaking down in harsh early Earth conditions. Some mineral surfaces even showed a remarkable ability to sort molecules by their “handedness” (chirality), a critical feature of life’s chemistry. By bringing these molecules together and organizing them into structured clusters, minerals may have transformed a chaotic “prebiotic soup” into ordered pockets of molecules ready to form the first complex systems of life. Though testing these ideas in the lab remains challenging, as shown by Bullard’s experiments, the notion that Earth’s minerals were more than passive bystanders in life’s origin story continues to captivate scientists.
Amino Acid chirality on Earth Vs Meteorites
Hazen and Sverjensky (2010) suggest we consider the complex puzzle of amino acid selection in the context of life’s origins, drawing on evidence from meteorites. They note that over 70 different amino acids have been identified in carbonaceous chondrites like the Murchison meteorite, featuring a mix of both left-handed (L) and right-handed (D) forms, with some studies indicating a slight excess of L-amino acids in certain cases. In stark contrast, life on Earth relies on just 20 specific amino acids, nearly all in their left-handed form, a pattern of selectivity also seen in life’s sugars, lipids, and polycyclic molecules. This raises a critical question: how did the early Earth select and concentrate this narrow subset of molecules from the diverse molecular inventory delivered by meteorites? Their work points to the potential role of mineral surfaces in facilitating this molecular sorting and organization, setting the stage for the emergence of life’s unique biochemistry.
Why does life use exclusively left-handed amino acids (L-AAs) when meteorites deliver a near-even mix of L and D forms?
The Meteorite Data vs. Life’s Bias
The Murchison meteorite, a treasure trove from space, reveals a fascinating snapshot of early chemistry. Analysis shows it contains a near 50/50 mix of left-handed (L) and right-handed (D) amino acids, though some studies suggest a slight excess of L-amino acids—about 1% more than D (Pizzarello & Cronin 2000). This balance hints at the raw materials delivered to early Earth. In stark contrast, life on Earth today is strikingly one-sided: nearly 100% of its amino acids are L-forms, with rare exceptions like D-amino acids found in bacterial cell walls. This dramatic shift from a mixed cosmic supply to life’s exclusive preference raises a compelling question: how did this molecular bias emerge?
Main Hypotheses (Problems)
Scientists have proposed two leading ideas to explain this transition, though both face hurdles. One theory involves circular polarized light (CPL). This type of light, which spirals as it travels, might have interacted with amino acids in space
Research suggests that L-amino acids might absorb right-handed CPL slightly more than D-forms, potentially breaking down D-amino acids over time and leaving a small L-excess (around 1%). While this could have started a preference in space or on early Earth, the effect is weak and may not explain the full dominance of L-amino acids in life.
- Circular Polarized Light (CPL):
- L-AAs absorb RH-CPL slightly more → breaks D-AAs (weak effect: ~1% excess).
- Vester-Ulbricht Mechanism:
- Beta decay’s parity violation → slight L-bias (unproven for AAs).
- The Quantum Hydrogen Hexagonal-Lattice Idea:
- Cosmic chiral vortices → polarized UV + spin-polarized electrons → strong L-selection.
The second hypothesis, the Vester-Ulbricht mechanism, links the bias to beta decay—a process in radioactive elements where emitted particles defy a symmetry called parity. This asymmetry might have given L-amino acids a tiny advantage, but there’s no solid evidence yet that it shaped amino acids in nature. Both theories hint that a slight initial tilt toward L-forms could have been amplified, perhaps by mineral surfaces acting as catalysts, as suggested in earlier studies of prebiotic chemistry. Still, the leap from a 50/50 mix to life’s 100% L-bias remains an open question, inviting further exploration.
Step 1: Primordial Chiral Pump
The electron flow in daisy petal pattern leads to a vortex action within the center, the hexagon’s opening.
- The Hexagonal 6H Lattice Creates Vortices:
- RH/LH electron flows generate circularly polarized UV (CPL) in star-forming regions.


Step 2: Meteorite Delivery
- Murchison’s L-Excess: Traces of vortex-polarized CPL in parent body.
- Earth’s Amplification:
- Tidal pools + lattice-aligned clays concentrate L-AAs (Hazen 2001).
Step 3: Toxic D-AAs as “Grit”
- 6H Theory New Insight: D-AAs disrupt phase-coherent protein folding (like RH vortices in LH lattices).
Disclaimer: This information is being provided within the guidelines of Fair Use and is not intended to provide individual health care guidance.
Reference
Hazen RM, Sverjensky DA. Mineral surfaces, geochemical complexities, and the origins of life. Cold Spring Harb Perspect Biol. 2010 May;2(5):a002162. doi: 10.1101/cshperspect.a002162. Epub 2010 Apr 14. PMID: 20452963; PMCID: PMC2857174. https://pmc.ncbi.nlm.nih.gov/articles/PMC2857174/
Inaki M, Liu J, Matsuno K. Cell chirality: its origin and roles in left-right asymmetric development. Philos Trans R Soc Lond B Biol Sci. 2016 Dec 19;371(1710):20150403. doi: 10.1098/rstb.2015.0403. PMID: 27821533; PMCID: PMC5104503. https://pmc.ncbi.nlm.nih.gov/articles/PMC5104503/
Chiral Hydrogen Lattices as the Origin of Cosmic Structure, Lyman Forest Press, June 16, 2025, First Draft, https://www.dropbox.com/scl/fi/7zalipi9hcpto4h2f61ra/Chirality_6_15_2025.pdf?rlkey=fmb70fgb1vv4px7qm81hh54qt&st=2y22fjz0&dl=0
